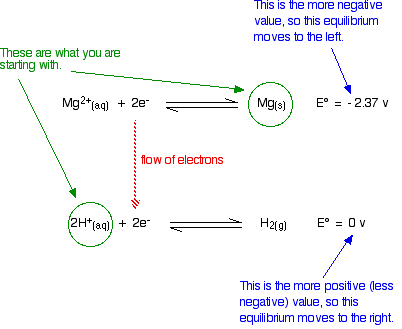
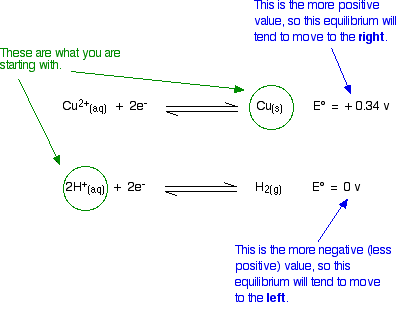
Predicting Reactions using Standard Electrode potentials
The half reaction with the more negative value will proceed to the left, and the other will proceed to the right. By looking at the standard electrode potentials for each half reactions, we can tell whether a reaction is possible.
- For example, in the case on the left, the reaction is possible because the Mg can become Mg ions and electrons from that can turn hydrogen ions into hydrogen gas
- In the case of the right though, the reaction cannot happen because the equilibrium will have to move to the opposite of what the standard electrode potentials are saying.
http://www.chemguide.co.uk/physical/redoxeqia/predict.html