Transition metals are elements in Group 3 to Group 11 of the periodic table, some of which are chromium, cobalt, copper, gold, and iron.
General Properties
Transition metals are metals. They are harder, more brittle and have higher melting points than the metals in Group 1 and Group 2. They have variable oxidation states and have colored ions.
The electrons in the d orbital are loosely bound, and moving from left to right, the 5 d orbitals get more filled. Transition metals have low ionization energies.
The electrons in the d orbital are loosely bound, and moving from left to right, the 5 d orbitals get more filled. Transition metals have low ionization energies.
d block and transition definition
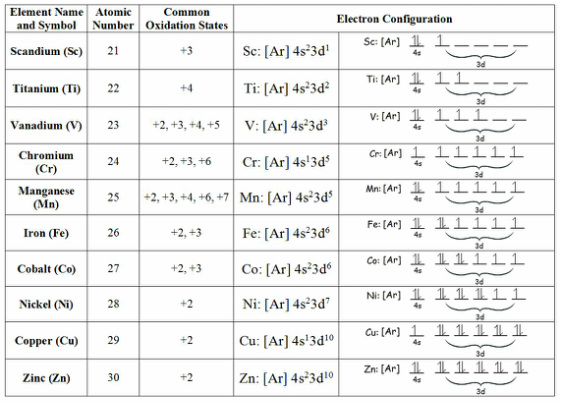
The elements on the periodic table that have 3d orbitals filling are called the d block elements. The first row of the d block starts with Scandium and ends with Zinc. Not all elements in the d block are transition metals. The definition of transition metals is a metal that forms one or more stable ions with incomplete d orbitals. From this definition Sc and Zn are not transition metals although they are part of the d block.
Electron configuration and trends
The electron configuration of the d block are as follows:
Sc[Ar] 3d14s2
Ti[Ar] 3d24s2
V[Ar] 3d34s2
Cr[Ar] 3d54s1
Mn[Ar] 3d54s2
Fe[Ar] 3d64s2
Co[Ar] 3d74s2
Ni[Ar] 3d84s2
Cu[Ar] 3d104s1
Zn[Ar] 3d104s2
Sc[Ar] 3d14s2
Ti[Ar] 3d24s2
V[Ar] 3d34s2
Cr[Ar] 3d54s1
Mn[Ar] 3d54s2
Fe[Ar] 3d64s2
Co[Ar] 3d74s2
Ni[Ar] 3d84s2
Cu[Ar] 3d104s1
Zn[Ar] 3d104s2
- Ionization energy increases from left to right across the row
- Higher oxidation states become relatively more unstable than lower oxidation states as you move from left to right
- The relative stability of +2 compared to +3 increases as you move from left to right
Variable oxidation states
Transitional metals have a range of oxidation states.
- Manganese for example can have oxidation states of +2, 3, 4, 6, and 7
- Common oxidation states for the first row transition elements are shown below:
Chemistry The Study of Matter Prentice Hall p.670
http://chemistry.about.com/od/elementgroups/a/transitionmetal.htmhttp://www.chemguide.co.uk/inorganic/transition/features.html
http://chemwiki.ucdavis.edu/Inorganic_Chemistry/Organometallic_Chemistry/Structural_Fundamentals/Periodic_Trends_of_the_Transition_Metals#Ionization_Energy
http://www.4college.co.uk/a/ss/varoxidation.php
http://chemwiki.ucdavis.edu/@api/deki/files/1220/(Remake)_Oxidation_States_for_First_Row_Transition_Metals.jpg